Scientific Information
Abstract
Background: The high number of people living with HIV in sub-Saharan Africa necessitates resource-efficient treatment approaches in order to ensure high quality long-term care. The VITAL trial investigates a viral load-driven differentiated service delivery model. The model triages patients into subgroups according to their viral load and clinical considerations. Patients with a suppressed viral load are monitored less intensively, thus freeing resources for patients with an increased viral load or other HIV-associated problems.
Methods: VITAL is a multicentre cluster-randomized controlled non-inferiority trial, enrolling persons who have been taking antiretroviral therapy at 18 nurse-led health care facilities (‘clusters’) in the districts of Butha Buthe and Mokothlong in northern Lesotho. The clusters are randomized in a 1:1 allocation to a control arm, receiving standard care, and an intervention arm. A viral load result-driven automated differentiated service delivery model (aDSDM) is implemented in intervention clusters. This aDSDM uses viral load results, other clinical characteristics and participants’ preference to automatically triage patients into groups requiring different levels of attention and care (see Figure 1 below). Additionally, two eHealth tools ensure that ensure an effective information flow: 1) Viral load results and other relevant information is sent directly to the participants' phones. 2) Healthcare providers will receive the results together with the recommended action directly on their study tablet. The primary endpoint is defined as: The proportion of participants engaged in care (defined as documented visit attendance) with documented viral suppression (<20 copies/mL) 24 months (16-28 months) after enrolment.
Discussion: The evidence generated in the VITAL trial intends to inform future national and international guidelines and policies. If successful, the VITAL aDSDM could be scaled up to the national level. Furthermore, evidence generated from nested studies may inform local/national policies.
Figures
Schematic overview. Determinants of follow up in VITAL.
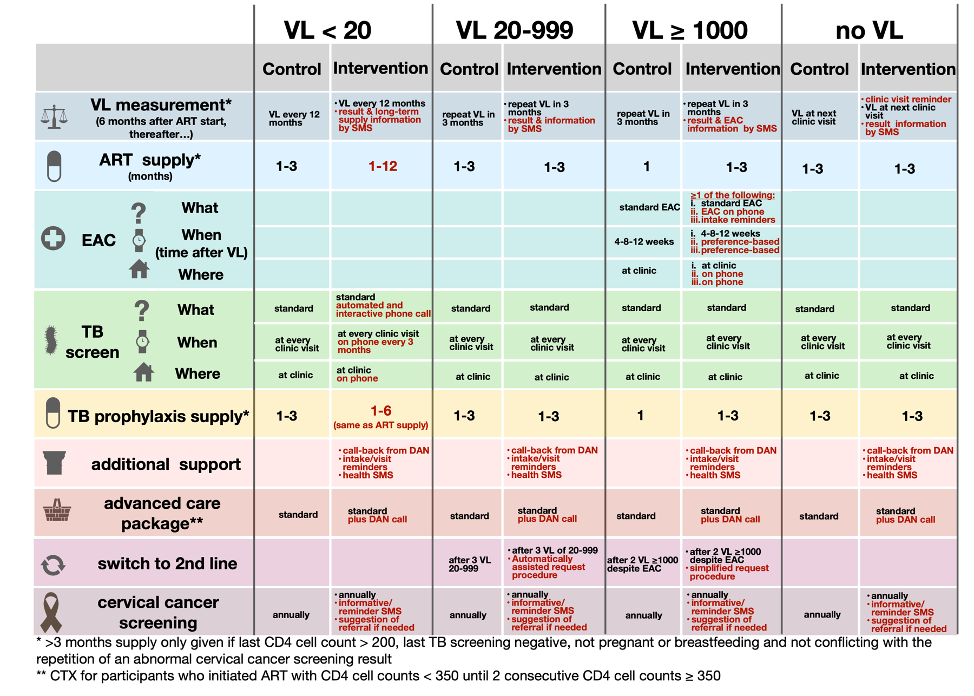
HIV care package and corresponding VITAL intervention. Red indicates main differences to standard of care.
Publications and further information
Trial registration
ClinicalTrials.gov: NCT04527874 (registered 27 August 2020).
Publications
Study protocol: PLoS One, 5 May 2022.
Pilot study: BMC Pilot and Feasibility Studies, 11 March 2022.